 
As white light passing through an element produced a spectrum with certain wavelengths of light missing. The emission spectrum on the right is that of helium.ġ) The most abundant hydrogen atom is composed ofĤ) What would happen if an electron orbited the nucleus in a circular path?ĥ) What did Bohr say about an electron orbiting the nucleus and the energy level it is allowed to have?Ħ) An electron can occupy orbits that areħ) What hypothesis did Bohr put forward to explain why the electron dose not lose energy and fall into the nucleus?Ĩ) What evidence did Bohr have to support his hypothesis?ĩ) By assuming the electron can act as both a particle and a wave what were scientists able to explain?īohr's model could also explain observed absorption spectra. He even managed to accurately predict previously undetected lines in the UV region of the spectrum of hydrogen. could move only in fixed orbits of specific energies.Įlectrons with low energy would orbit closer to the nucleus while electrons with high energy orbit further from the nucleus.īohr's model could accurately predict the lines appearing in emission spectra. orbit the nucleus without losing energy 
Isotopes of the same element have the same number of protons and electrons but differ in the number of neutrons found in their nucleus.īefore Chadwick's discovery of the neutron in 1932, a Danish scientist by the name of Neils Bohr refined Rutherford's model in 1913 by proposing that electrons: How could two atoms of the same element have identical chemical properties but one be radioactive and the other not? A British scientist Frederick Soddy who made this observation called varieties of the same element isotopes.Ĭhadwick was now able to explain the existence of isotopes through his discovery of the neutron. One observation was the existence of radioactive variances of the same element. With the discovery of the neutron three subatomic particles were identified that would help explain observations made at the atomic level. The particle proposed by Rutherford as having significant mass and no charge. In 1932 James Chadwick identified the neutron. 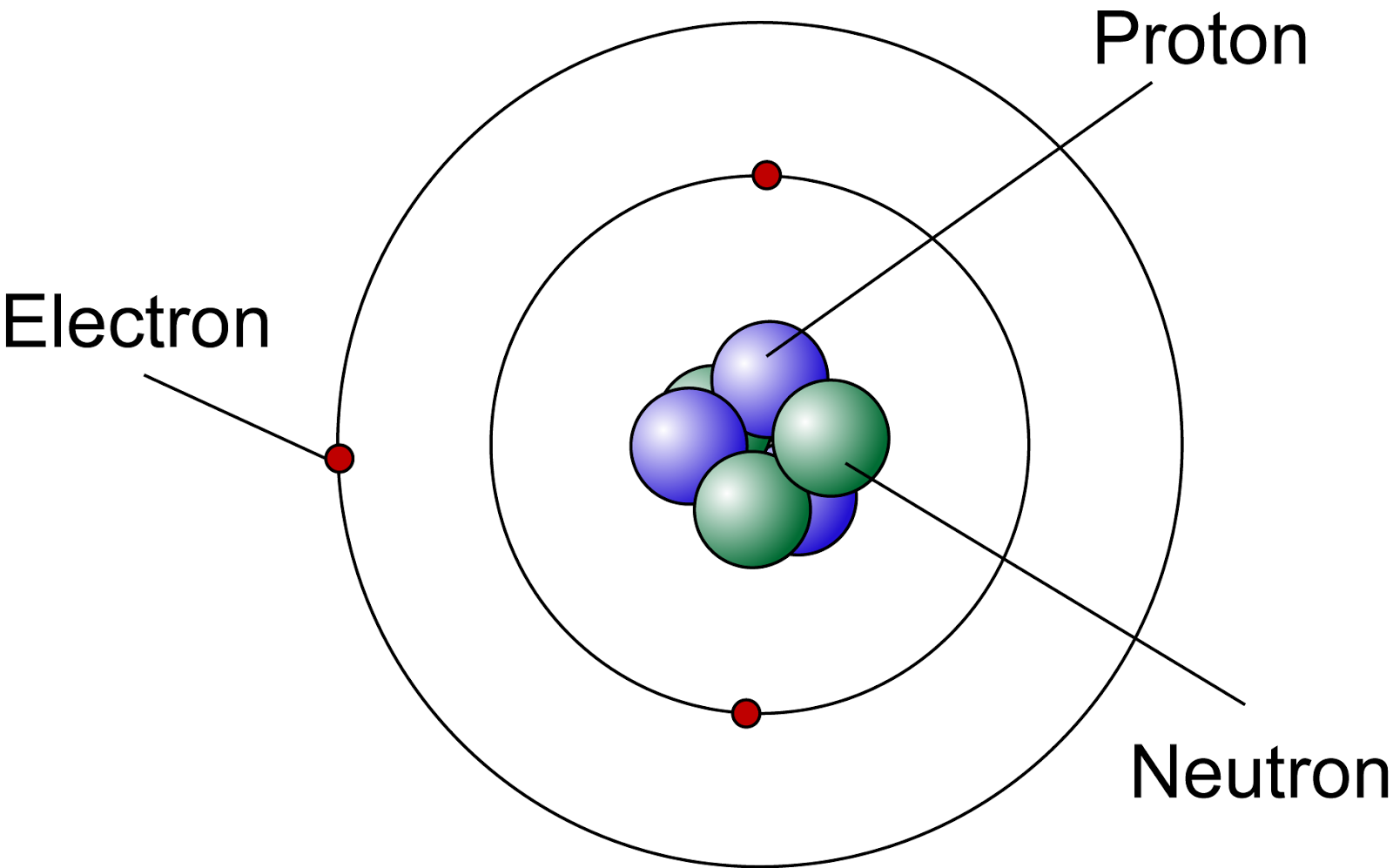
History of the atom-James Chadwick and Neils Bohr 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
